Flame spread over solid
fuel beds
Not proficient in English? Try the Slovak version.
INTRODUCTION
Flame
spread over thin solid fuels is a useful paradigm for studying the behavior of
more complex flame spread problems, e.g., in building fires. Our
experiments on flame spread in non-standard atmospheres have shown that the
unequal rates of diffusion of thermal energy and oxidizer (the "Lewis
number" effect) are important influences that have not received much
attention in prior studies. We have also discovered a new type of flame spread process in which the flame front is cellular
and explained this in terms of Lewis number effects in conjunction with
reactant interdiffusion near extinction. Our
theoretical modeling has confirmed the importance of Lewis number effects and
shown that such effects can be described via extensions of classical theory.
The
effects of buoyancy-induced convection and radiative heat transport on flame
spread over both thin and thick fuel bed materials have been studied.
Experiments have been conducted at earth gravity and at microgravity (µg) in
the drop towers at the NASA-Glenn Research Center in Cleveland, OH. Most
experiments were conducted in a 20 liter combustion
chamber (Fig. 1). Exactly the same apparatus was used for 1g and µg tests. In
most thin-fuel experiments 5 cm wide Kimwipe samples
15 cm long were used and were held by aluminum quenching plates. Thick-fuel
samples studied have included polystyrene, polyurethane, polyphenolic
and carbon foams. The samples were ignited by an electrically-heated
Kanthal wire. The flame spread
process was imaged via three video cameras and a laser shearing interferometer.
The
objectives of these experiments are to study the effect of diluent type, and
particularly the radiative properties of the diluent gas, on flame spread rates
and extinction conditions. Our current
state of understanding of these topics is discussed below.
EFFECT OF DILUENT TYPE
The
effect of inert gases He, Ar, N2, CO2
and SF6 on flame spread over thermally-thin fuels were tested since
these inerts provide a variety of radiative
properties and oxygen Lewis numbers. For He, N2 and Ar diluents, the flame spread rates (Sf)
at µg were always lower than the 1g values and the minimum oxygen
concentrations that would support flame spread were lower at 1g than µg. These
findings are consistent with prior studies in O2-N2 atmospheres
and results from the greater radiative heat losses at µg due to the increased
flame thickness (d) at µg. In contrast, for CO2 diluent,
Sf was slightly higher at µg and the
minimum O2 concentration was lower (Fig. 2). For SF6
diluent, Sf was substantially
higher at µg for all oxygen concentrations and the minimum O2
concentration was significantly lower (Fig. 2). These effects were
attributed to changes in the character of radiative transfer for CO2
and SF6. He, Ar and N2 diluents
do not emit thermal radiation and thus only the H2O and CO2
combustion products radiate significantly. For our test conditions tested the
Planck mean absorption length (LP) of the combustion products in
these diluents is typically 1 m, which is much larger than d, consequently,
radiative transport is optically thin. However, for CO2 and
especially SF6 diluents, LP is comparable to d or smaller, thus
reabsorption effects cannot be neglected. With reabsorption, radiation emitted
near the flame is not lost to the surroundings and instead augments
conventional thermal conduction to increase Sf
above radiation-free values. Interferometer images (not shown) showed that d increased at µg for all
diluents but most dramatically for radiatively-active
diluents, most likely due to these reabsorption effects.
Experiments on flame spread over thermally-thick fuels have been conducted using foam fuels
to obtain low density and thermal conductivity, and thus large spread rate (Sf) over thermally-thick fuels compared to dense
fuels such as PMMA. This scheme enabled
meaningful results to be obtained even in 2.2 second drop tower
experiments. It was found that, in
contrast conventional understanding; steady spread can occur over thick fuels
in quiescent microgravity environments, especially when a radiatively active
diluent gas such as CO2 is employed (Figs. 3, 4). This is proposed to be due to radiative
transfer from the flame to the fuel surface that can lead to steady spread even
when conductive heat transfer from the flame to the fuel bed is
negligible. Radiative effects are more
significant at microgravity conditions because the flame thickness is larger
and thus the volume of radiating combustion products is larger at microgravity. The effects of oxygen concentration and pressure
are shown and the transition from thermally-thick to
thermally-thin behavior with decreasing bed thickness is demonstrated. Radiative flux measurements confirm the
proposed effects of diluent type and gravity level. These results are particularly noteworthy
considering that the International Space Station employs CO2 fire
extinguishers; our results suggest that helium may be a better inerting agent on both mass
and mole bases at microgravity even though CO2 is much better on a
mole basis at earth gravity.
|
|
|
|
Figure 1. Schematic of drop
frame and interferometer apparatus. The fuel bed is mounted inside the
chamber parallel to the plane of the page. |
Figure 2. Flame spread
rates vs. O2 mole fraction at 1 atm in
helium, carbon dioxide and sulfur hexafluoride diluents. |
|
|
|
|
Figure 3. Effect of oxygen concentration on spread
rates over thick solid fuel beds at µg and earth gravity. |
Figure 4. Effect of pressure on spread rate over thick
solid fuel beds at µg and earth gravity (polyphenolic
foam, density 0.0267 g/cm3, atmosphere 40% O2 - 60% CO2.). |
FLAME SPREAD
IN PARTIALLY-PREMIXED ATMOSPHERES
Fires in
enclosures usually burn in under-ventilated conditions, leading to atmospheres
containing unburned fuel or intermediates such as CO. With this motivation, thin-fuel
flame spread experiments were conducted at 1g and g in atmospheres containing
sub-flammability-limit concentrations of gaseous fuels in O2-N2
atmospheres. CO and CH4 were used for the gaseous fuels in partially-premixed atmosphere tests, plus H2, C3H8
and NH3 for 1g tests only. 1g tests showed that that for some fuels
such as CO and H2, there is a substantial effect of gaseous fuel on Sf, whereas for other fuels such as NH3,
there was practically no effect. Remarkably, for CO fuel (Fig. 5), a very important
case for practical applications, Sf was
higher and the minimum O2 concentration was lower when a given
number of oxygen atoms in the ambient atmosphere was present in the form of CO
rather than O2. Moreover, these data do not even account for the fact
that in practical fires the vitiated air will be hotter than ambient due to the
heat release associated with the partial oxidation. For all gaseous fuels
enhancement of Sf correlated well with the
characteristic chemical reaction rate of the premixed fuel and no correlation
with the heating value or diffusive properties of the premixed fuel was
observed. In the current flight definition study, this work will be extended to
thick fuels using longer-duration g experiments.
It was found
that the effect of adding gaseous fuel to the ambient atmosphere was
qualitatively similar at 1g and g but the effect is stronger at g than 1g, and
in fact Sf is actually higher
at g than 1g at high premixed fuel concentrations (Fig. 6). Also, the effect of
added gaseous fuel was found to be more substantial at higher oxygen
concentration and with CO fuel. All of these results are consistent with the
simple theoretical model proposed by the PI which shows the effect of the
premixed fuel is to cause a partially-premixed flame sheet to occur upstream of
the conventional non-premixed flame. This additional flame increases the total
heat flux to the fuel bed and thus Sf. Finite-rate chemistry of the
premixed flame was found to affect the additional heat flux, even when the
nonpremixed flame is at the high Damkohler number
(mixing-limited) condition.
|
|
|
|
Figure 5. Effect of O2
mole fraction and gaseous CO addition on Sf. Solid lines and data
points: experiment; dashed lines: theoretical predictions. f is the equivalence ratio of the
premixed atmosphere. |
Figure 6. Measured and
predicted flame spread rates vs. f. Atmosphere: 18% O2 in N2
at 1 atm with CO as added gaseous fuel. |
CONCURRENT-FLOW
FLAME SPREAD
A study of
upward flame spread over solid fuels was conducted to clarify the mechanisms of
spread rates for concurrent-flow flame spread and in particular, buoyancy
effects on this process. It was proposed that, contrary to many prior
theoretical predictions, upward flame spread could be steady because convective
losses to the sides of the fuel samples and/or surface radiative losses prevent
the flame length and thus spread rate from growing indefinitely. These losses
were argued to be unavoidable because the flame length will grow until these
losses balance the heat generation rate. Scaling relations for the spread rates
in the presence of convective and radiative losses, laminar and turbulent flow,
buoyant and forced convection, and thin and thick fuels were derived. Tests of
some of these relations were conducted for upward-propagating flames over tall,
thermally-thin fuel samples, subject to buoyant
convection only, for a range of pressures, oxygen mole fractions, diluents and
fuel bed thicknesses. In this manner a seven-decade range of Grashof number, defined as gW3/n2, where g is the
gravitational acceleration, W the fuel bed width and n the kinematic
viscosity, was studied. Only conditions away from quenching were studied to
minimize chemical influences. Flames were found to achieve steady values of
both Sf and flame length when the
sample was sufficient tall. Measured values of Sf,
normalized by the opposed-flow (downward) spread rate with the same atmosphere
and fuel bed (Sf,opp),
are shown in Fig. 7. At low GrW, Sf/Sf,opp
~ GrW1 with the value of the proportionality constant
being slightly different for different atmospheres. At higher W, Sf is independent of GrW,
indicating a transition to radiatively-stabilized
flame spread. At intermediate GrW, there
is some indication of a region where Sf/Sf,opp ~ GrW4/7
as would be characteristic of turbulent buoyant regime. The data deviate from Sf/Sf,opp
~ GrW1 behavior towards Sf/Sf,opp ~ GrW4/7 near GrW = 20,000, which is close to where a
predicted transition from laminar to turbulent behavior occurs. Furthermore, visually
the flames were observed to change to a turbulent structure near this value of GrW.
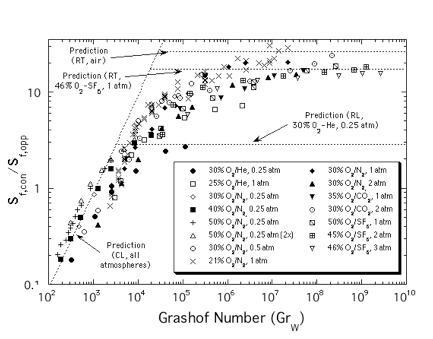
Figure 7. Effect of Grashof
number (GrW) on upward flame spread rate,
normalized by downward spread rate, over thin fuel samples.
PUBLICATIONS
Greenberg, J.
B., Ronney, P. D., "Analysis of Lewis Number Effects in Flame
Spread," International Journal of Heat and Mass Transfer, Vol. 36,
pp. 315-323 (1993).
Zhang, Y.,
Ronney, P. D., Roegner, E., Greenberg, J. B.,
"Lewis Number Effects on Flame Spreading Over Thin Solid Fuels," Combustion
and Flame, Vol. 90, pp. 71-83 (1992).





